ISO 13485 quality management
Device lifecycle files, supplier controls, CAPA evidence, and production release records are organized for quality-system review.
Agilent teams document throughput, calibration, reagent handling, and connectivity in formats that biomedical engineering, laboratory directors, and regulatory affairs can inspect without interpretation gaps.
| Analytical Workflow | |
|---|---|
| Primary systems | LC/MS, GC/MS, HPLC, clinical chemistry, molecular sample preparation, and controlled consumable workflows. |
| Validation records | Linearity, precision under CLSI EP05, method comparison, carryover studies, and calibration interval evidence. |
| Connectivity | |
| LIS integration | HL7 v2.x, ASTM E1394, middleware mapping, bidirectional order/result messaging, and audit trails for result release. |
| Data integrity | Role-based access, electronic signatures, time-stamped result review, and controlled export for CAP and CLIA inspections. |
| Service Control | |
| Uptime program | Preventive maintenance windows, parts pre-positioning, remote diagnostics, and escalation routing for critical assays. |
| Compliance files | Certificates, IFU packs, lot traceability, software revision records, and change-control notices. |
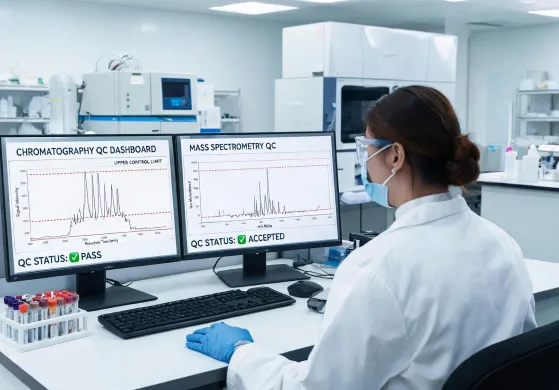
Structured validation packs connect sample handling, reagent lots, calibration, and QC trend review. The result is a defensible chain of evidence for high-complexity laboratories that need repeatable performance across shifts and sites.

Agilent implementations map orders and results into existing LIS environments while preserving auditability. Interfaces can be scoped for Epic Beaker, Oracle Health PathNet, middleware engines, and regional reporting portals.

Service contracts include preventive maintenance schedules, documented parts replacement, application specialist access, and lifecycle planning so value-analysis teams can compare downtime risk instead of just acquisition cost.
Device lifecycle files, supplier controls, CAPA evidence, and production release records are organized for quality-system review.
Assay documentation, intended use records, performance summaries, and risk notes help teams prepare jurisdiction-specific dossiers.
QC trending, calibration evidence, proficiency testing records, and change-control logs support high-complexity lab inspections.
Software revision control, vulnerability response timing, and SBOM request workflows help IT security teams approve networked instruments.
Share your sample volume, LIS environment, assay scope, and service constraints. An applications specialist can map the validation path and supporting documents before your team enters value analysis.